Understanding Biomolecular Interfaces
Uncovering the molecular rules that govern protein behaviour at interfaces
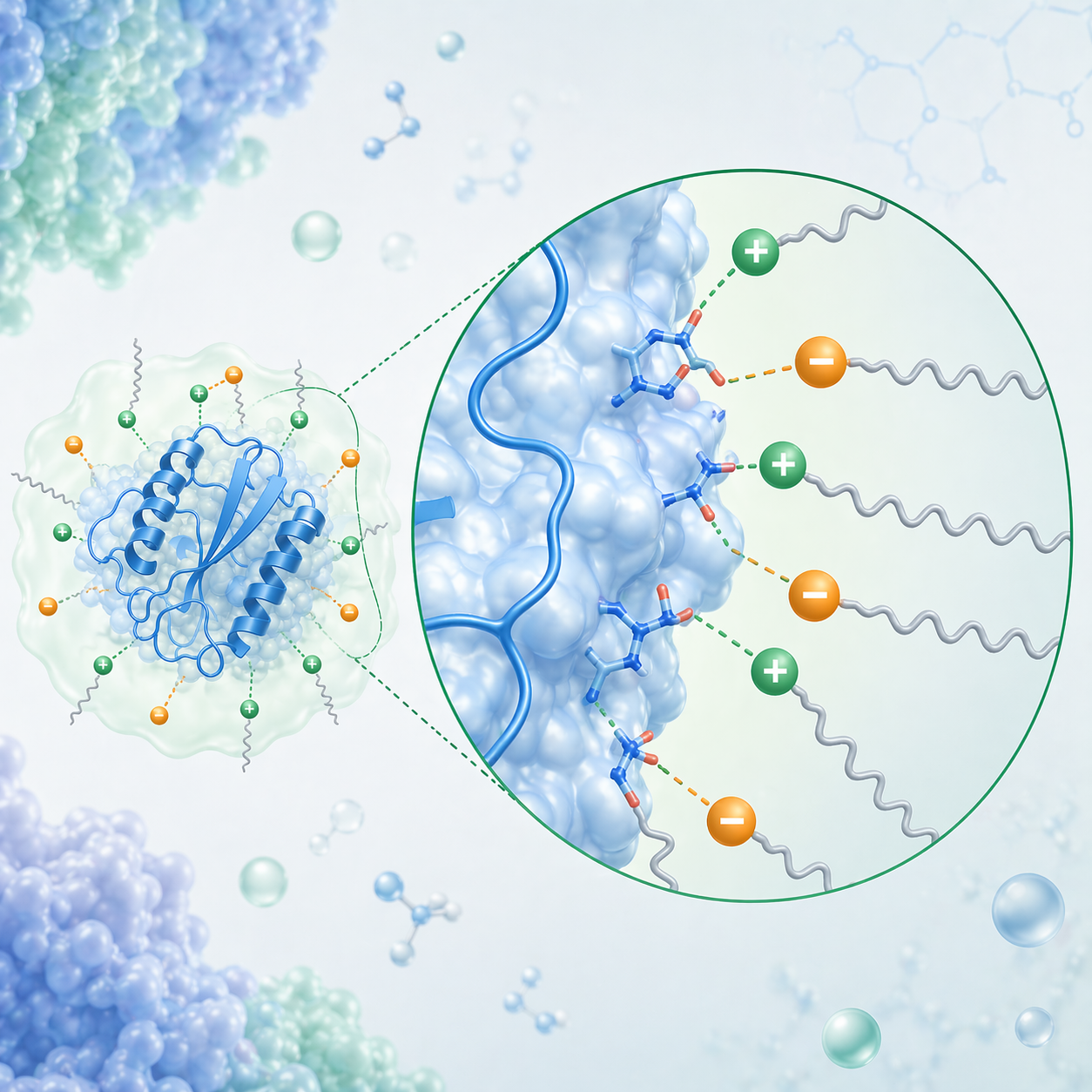
Proteins are highly sensitive to their surrounding environment, and their stability and function are often governed by interactions at biomolecular interfaces. In non-native environments, such as formulation systems or biological barriers, these interfacial interactions can lead to protein destabilisation, aggregation, or loss of activity, limiting the effectiveness of protein therapeutics.
Our work has focused on uncovering how specific intermolecular interactions and structural features at the protein surface govern these behaviours. We have shown that protein modification is not uniformly distributed, but is dictated by tertiary structure, residue accessibility, and the local physicochemical environment, linking protein structure directly to interfacial reactivity and functional outcomes. Building on this mechanistic understanding, we have investigated protein–surfactant and protein–polymer interactions, demonstrating how strategies such as hydrophobic ion pairing (HIP) can be used to modulate protein surface properties and control solubility, stability, and activity. Through systematic studies, we have established that these interactions are not simply formulation tools, but fundamental drivers of protein behaviour in complex environments.
We have also demonstrated how interfacial design strategies can directly influence biological performance, including enhanced stability and activity through macromolecular and supramolecular conjugation approaches, and how these principles extend to complex biological systems. In parallel, our work has contributed to defining this area more broadly through the study of artificial lipidation as a strategy for biomolecular interface engineering, highlighting how both covalent and supramolecular approaches can be used to modulate protein behaviour across scales, from molecular structure to therapeutic performance.
Together, this work establishes a mechanistic framework for understanding biomolecular interfaces, revealing how protein behaviour can be rationally tuned through control of surface interactions. This foundation underpins the development of predictive tools and engineered systems for next-generation therapeutic applications.
Related Publications
-
Artificial lipidation of proteins and peptides: From mechanism to clinical applicationsRead More
J Mu, E Vong, S Carmali. The FEBS Journal, 293(5), 1269–1284 (2026). -
Re-engineering lysozyme solubility and activity through surfactant complexationRead More
J Mu, L Mao, GP Andrews, S Carmali. Materials Advances, 5(21), 8515–8523 (2024). -
Chemical zymogens for the protein cysteinomeRead More
MC Montasell, P Monge, S Carmali, LM Dias Loiola, DG Andersen, et al. Nature Communications, 13(1), 4861 (2022). -
Dimerization of the peptide CXCR4-antagonist on macromolecular and supramolecular protraction arms affords increased potency and enhanced plasma stabilityRead More
M Harms, RF Hansson, S Carmali, Y Almeida-Hernández, et al. Bioconjugate Chemistry, 33(4), 594–607 (2022). -
Tailoring site specificity of bioconjugation using step-wise atom-transfer radical polymerization on proteinsRead More
S Carmali, H Murata, K Matyjaszewski, AJ Russell. Biomacromolecules, 19(10), 4044–4051 (2018). -
Tertiary structure-based prediction of how ATRP initiators react with proteinsRead More
S Carmali, H Murata, E Amemiya, K Matyjaszewski, AJ Russell. ACS Biomaterials Science & Engineering, 3(9), 2086–2097 (2017).
